 
The three hybrid orbitals remain in one plane and make an angle of 120° with one another.Moreover, sp² hybridization forms trigonal symmetry and is maintained at 1200.The process of sp² hybridization involves mixing one 's' orbital and two 'p' orbitals to form sp² hybridization.sp² hybridization is also called diagonal hybridization similarly, sp² hybridization is called trigonal hybridization.Thus, the new orbitals formed are called sp² hybrid orbitals. When one 's' and two 'p' orbitals of the same shell of an atom mix to form ² equivalent orbitals, sp² hybridization occurs. All compounds of carbon-containing triple bonds like C2H2.All compounds of beryllium like BeF2, BeH2, BeCl2.Further, each sp hybridized orbital has 50% s and 50% p character, thus making it equal.sp hybridization is also called diagonal hybridization.The newly formed sp hybridized orbitals form linear molecules with an angle of 180°. 
sp hybridization usually involves mixing one 's' orbital and one 'p' orbital of the same energy to produce a new hybrid orbital known as an sp hybridized orbital.When one 's' and one 'p' orbital in the same main shell of an atom mix to form two new equivalent orbitals, it is called sp hybridization. The points below discuss the different types of hybridization with examples. The hybridization can be classified into sp³, sp², sp, sp³d, sp³d², sp³d³, based on the types of orbitals engaged in mixing. Give the hybridized states of each of the carbon atoms in the given molecule. The hybridization of the molecule can predict the molecule's shape.In contrast, the smaller lobe on the opposite side has a negative sign. The more prominent lobe of the hybrid orbital constantly has a positive sign.Hybridization occurs exclusively during the bond formation and not in an isolated gaseous atom.The number of hybrid orbitals formed equals the number of atomic orbitals mixing.Further, the atomic orbital has the same energies that undergo hybridization.Both half-filled and full-filled orbitals can be involved in the hybridization process.Hybridization gives rise to a new type of hybridized orbitals.Let us see the key takeaways from hybridization: 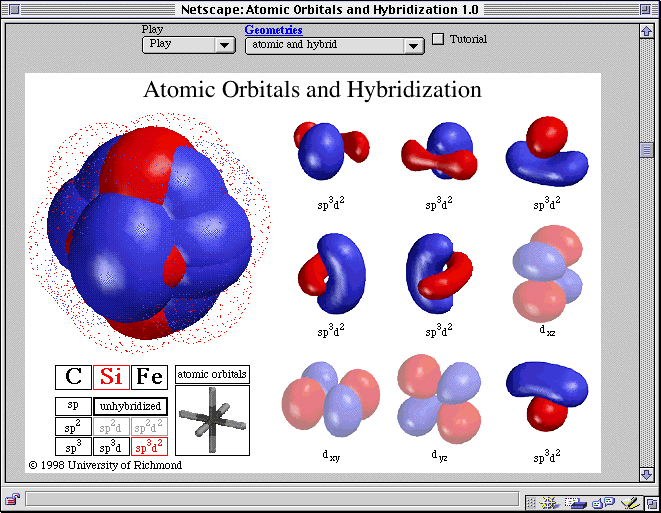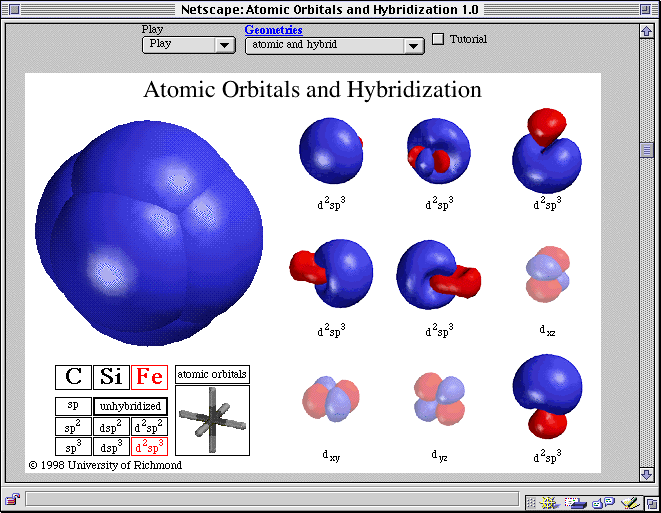
Hence, mixing different orbitals creates a new orbital known as hybrid orbitals. Generally, two 'p' orbitals or two 's' are mixed, or a single 'p' orbital with 's' orbital are mixed, or the mixing of 's' orbital with 'd' orbital happens. Nonetheless, the hybrid orbital is used to explain atomic bond properties and molecular geometry.Īdditionally, hybridization involves atomic orbitals of similar energies that are mixed. Hybridization is the process that involves the redistribution of energy of orbitals of each atom to produce orbitals of equivalent energy that happens when two or more atomic orbitals combine to form a hybrid orbital in a molecule. Both the filled and half-filled orbitals can be involved in the hybridization process. Moreover, the atomic orbitals of similar energy levels are hybridization. Further, there is a formation of hybrid orbitals having entirely different energies, shapes, etc., due to the intermixing. In chemistry, hybridization is recognised as mixing two atomic orbitals to rise to a new type of hybridized orbitals. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
